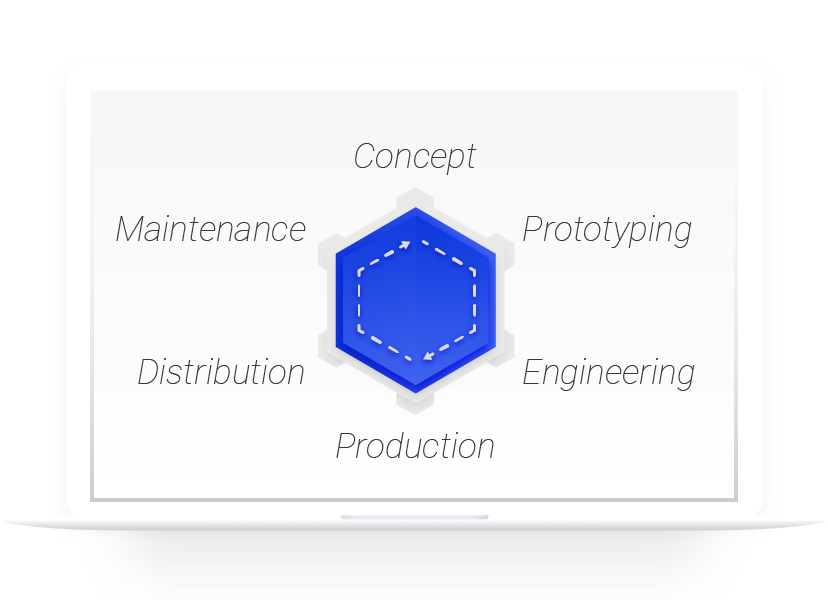
Digitalisation starts with PLM
Highstage has helped create top-performing businesses for more than 15 years. Aligning structure, processes, and resources. Highstage aids you in streamlining your business and boost productivity. Accenture claims that PLM (Product Lifecycle Management) is the best investment to fuel digitalization for any business. Highstage is the fastest way to reap the benefits. Try it today.
Learn how PLM can fuel your digitalizationAdvantages of Highstage
By using Highstage you should expect considerable increases in productivity
One size does NOT fit all
At Highstage, we do believe that every company is different, and that every company needs a different solution. Consequently, we do not move to centralized cloud solutions where everyone is handled with the same tools and formats. We offer all customers a highly customizable platform, which they can customize themselves, to adapt Highstage as an optimal solution exactly for them.
Industries
We have successfully served a variety of industries to create better products.
Consumer Electronics
Delivering complex solutions to the market is challenging in today’s global marketplace. Maintaining the supply chain updated is critical to eliminate delays, faults and getting products to market first. Highstage was created to help electronic companies deliver products under such demands and getting an iron grip of the Finished Products, Metadata and Bill of Materials.
Medical devices
Medical device companies encounter several challenges to successfully deliver innovative and reliable products while complying with MDR, FDA and ISO quality system regulations. Paper-based systems and Traditional file-centric document management tools fail to provide the full design record and at the same time slows the pace of development. Highstage allows the organisation to act faster by getting full control over relevant documentation, relation to the product parts.
Telecom & IoT
Telecom and IoT-services are becoming ubiquitous. The underlying devices driving this are getting more complex and adds further complex design challenges than ever before. Highstage has provided PLM, PDM and DMS tools for this industry of more than 20 years, and counting. Therefore Highstage is the perfect partner for an efficient and high-quality solution that helps solve engineering and supply-chain challanges in modern development environments. This allows companies to concentrate on delivering state of the art products and turn ideas into the next breakthrough in connected devices.
Other industries
Energy - Asset management - Military and Defense - Engineering - Software
Finally: Proper PLM for Small and Medium Enterprises
Don't believe what you've been told. Highstage is the PLM system even for small companies. Both the start-up friendly pricing, and the simple and incremental deployment, makes it a smart choice for all types and sizes of companies. This is what our customers say about us. Find more customer cases and testimonials under the industries.
"Highstage paid for itself quickly"

We save approximately one manhour week for every small project we run, in procurement activities only. Even though we ran a huge and intensive deployment project, it all paid itself in an approximately a year.Geir Berge (Procurement manager, Dwellop AS)
"The benefits are endless"
Basically, Highstage works as a data management level for the entire organization, and in combination with existent IT infrastructure where Highstage combines all together or as stand-alone. We have added a number of extra features, such as a Statistics Module for gathering and analysis of data from test systems in our production, Supply Chain link to our biggest customers and suppliers, Dynamics-AX Viewer for non-Dynamics-AX users.

The benefits are endless. The argument that you don’t have the time to implement Highstage and to understand the values form the platform, is something that needs to be challenged. It is, without doubt, the best management system on the market.Claus Dall Hansen (CEO and co-founder, Combilent A/S)